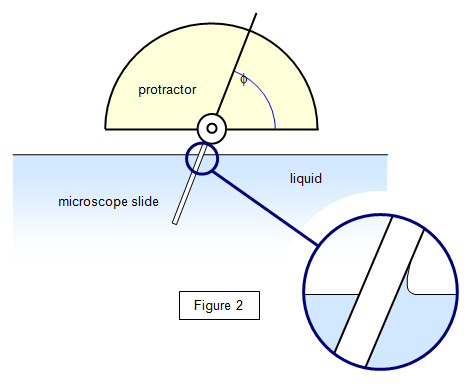

When a liquid surface meets a solid surface the angle
(f) between the two surfaces is important.
This angle is
known as the angle of contact, and its size determines whether the liquid will spread over
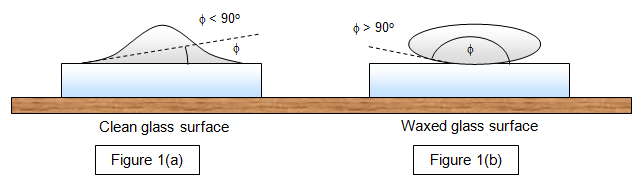
the surface or whether it will form droplets on it. If φ is less than
90o the liquid will spread and wet the surface (Figure 1(a)), while if it is greater
than 90o it will form droplets (Figure 1(b)). This effect can be seen clearly with
water on glass. For a clean glass-water boundary the angle of contact is nearly zero but this
increases to well over 90o if the surface is waxed.
In fact a surface will be wetted by water if the angle of contact is less than 90o, and so waterproofing is designed to produce an angle of contact that is greater than 90o. Addition of soap to water will reduce the angle of contact and is therefore useful in washing!