USES OF RADIOACTIVE ISOTOPES
Radioactivity can be used in a number
of ways. Some of these are described below.
1. Radioactive dating
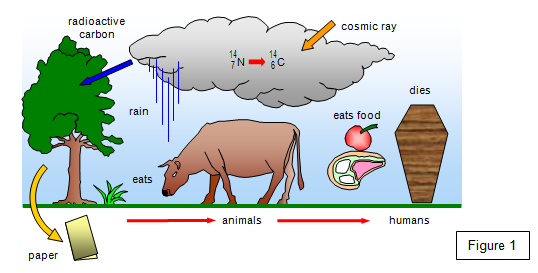
(a) CARBON 14 -
for dating material that has been alive or has been made from once living material. This is used for
dating wood, bones, Egyptian mummys, the Dead Sea scrolls, old paintings etc.
(b) URANIUM 238 or
POTASSIUM 40 for dating rocks. Used because of their very long half lives
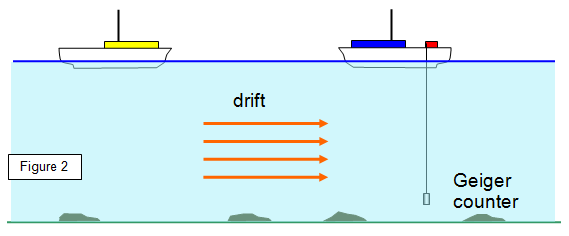
2.
Radioactive tracers
If a little radioactive material is put into a moving liquid the path of this
liquid can be tracked. Used in testing blood flow, tracking underground streams and following the
movement of silt in rivers
4. Thickness gauge
A beta source is put on
one side of a sheet of material and a Geiger counter on the other. The amount of beta radiation
that gets through the sheet will give you an idea of its thickness.
5. Cracks in
castings
A gamma source is placed in a metal casting and a Geiger counter moved over its
surface. If there are any cracks in the metal gamma radiation can get through and be
detected.
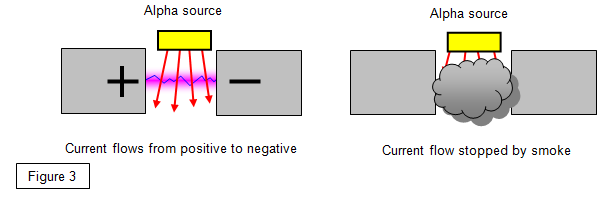
6. Smoke alarms
Many houses have a smoke alarm using a weak alpha
source. When smoke gets into the detector the alpha particles cannot get through to the sensor
and the alarm goes off.
6.
Sterilisation of food
Bacteria in food can be killed if exposed to gamma radiation.
7.
Medical uses of radioisotopes
These fall into two main sections:
(i) diagnostic – where the
radiation is used to locate a problem
(ii) therapeutic – where the radiation is used to treat a
problem
(a) Gamma radiation from Cobalt 60 has been found to be very useful in the
treatment of some types of cancer
(b) Syringes and other medical equipment can be sterilised
using gamma radiation
(c) Certain radioisotopes called tracers will concentrate in specific
organs for analysis – for example iodine 131 in the thyroid and technetium 99 in the brain, lungs,
and red blood cells and can then be used to diagnose defects
(d) Iodine 131 is also used to
treat thyroid cancer (it has a half life of just over 8 days)
(e) Iodine 123 (half life 13 hours) is
also injected into a patient where it concentrates in some organs and the gamma radiation emitted
by the source is scanned with a camera.
A VERSION IN WORD IS AVAILABLE ON THE SCHOOLPHYSICS USB