

Marie Curie thought that the strength of a radioactive source
and therefore the number of particles or gamma rays that it emitted every second did not change
with time but in fact she was wrong. The strength gets weaker as time goes by. Every time the
source emits a particle its activity decreases. There will be fewer radioactive atoms and more
stable ones.
The following graph shows how the activity of a source (in counts per second)
decreases with time.
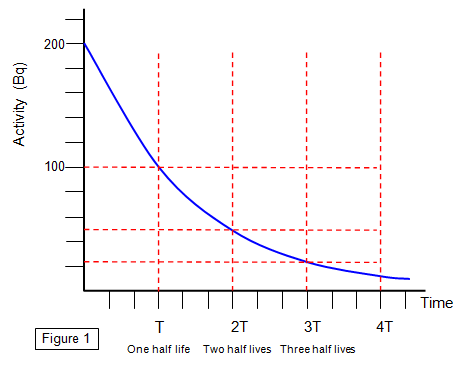
It's very difficult to
decide when a source has lost ALL of its radioactivity but the time for it to drop to HALF its original
value can be found easily.
The average time taken for the activity of a sample of
radioactive material to drop to HALF its original value is called the HALF LIFE of the source
(T).
This half-life depends only on the material of the source and different radioactive
isotopes have different half lives.
This means that if we have some
material that has a half life of 20 days then after 20 days it will only be HALF as radioactive as it
was at the start, after another 20 days, i.e 40 days after the start it will only be a quarter as
radioactive, one eight after 60 days and so on.
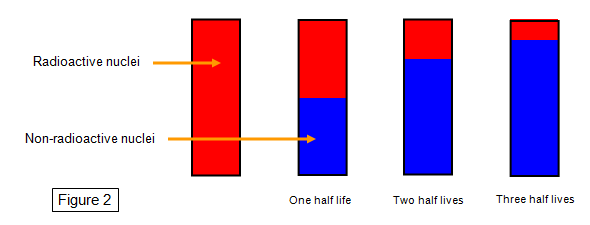
We can show this in another way by the
following diagram. If we start with a sample of material where all the nuclei are radioactive and
then watch how this changes as time goes by we will see that the number of radioactive nuclei
gets less while the number of non-radioactive nuclei gets greater. The total number of nuclei in
the sample stays the same.

| Material | Half life |
| Carbon 14 | 5700 years |
| Uranium 238 | 4500 million years |
| Radium 226 | 1600 years |
| Plutonium 239 | 24 000 years |